Yes, evidence is mounting that the Pfizer and Moderna vaccines cause similar adverse reactions. U.S. regulatory officials were alerted to this fact in December 2020, and since then, more research has been conducted to further investigate the potential side effects of both vaccines.
Marks said the FDA was aware of reports of CVST and thrombocytopenia in people who had received the vaccines, but that it had not been able to confirm any cases. He added that the agency was continuing to investigate these reports and would update its guidance if necessary.
The FDA has previously said that it is monitoring for potential safety issues related to the vaccines, including rare blood clotting disorders such as CVST with thrombocytopenia. The agency has also urged healthcare providers to report any suspected adverse events following vaccination.
Mark’s statement is not supported by the evidence presented in the news reports, recent studies, scientist’s warning, and data from the Vaccine Adverse Event Reporting System (VAERS). The evidence suggests that there are potential risks associated with vaccines that should be taken into consideration when making decisions about vaccination.
Study released today links Pfizer, Moderna to blood clots
The study, which was conducted by researchers at Oxford University and published in the British Medical Journal, found that the risk of developing CVST blood clots after receiving a COVID-19 vaccine was similar for all three approved vaccines in the European Union. The study looked at data from more than 17 million people who had received either the Pfizer/BioNTech, Moderna or AstraZeneca vaccines.
The researchers concluded that there was no significant difference in the risk of developing CVST blood clots between any of the three vaccines. They also noted that while there were some cases of CVST reported after vaccination, it is important to remember that this is a rare condition and that the benefits of vaccination far outweigh any potential risks.
The study found that the risk of CVST was very low for all three vaccines. The risk of CVST was 4 in 1 million people for the Pfizer and Moderna vaccines, and 5 in 1 million people for the AstraZeneca vaccine. The study also found that the risk of CVST was higher among younger adults than older adults, with a rate of 8 in 1 million people for those aged 18-49 compared to 2 in 1 million people for those aged 50 and over.
The researchers concluded that the risk of blood clots following COVID-19 vaccination is real and should be taken into account when considering the benefits and risks of vaccination.
- “These findings are consistent with what we know about how vaccine-induced spike proteins can on their own cause cell signaling through interactions with the ACE-2 receptors,†said Lyn Redwood, RN, MSN, president emerita of Children’s Health Defense. “When this happens, it can result in inflammation and a host of other potentially pathological events in the epithelial lining of the blood vessels which can then trigger pro-inflammatory cytokines capable of activating coagulation systems and down-regulating anticoagulant pathways resulting in clot formation.â€
The study found that thrombocytopenia, a condition in which the body has an abnormally low number of platelets, is a rare but possible side effect of both the Pfizer and Moderna vaccines. The authors noted that this side effect is likely to be more common in people with underlying medical conditions such as diabetes or kidney disease. They also suggested that healthcare providers should monitor patients for signs of thrombocytopenia after vaccination and take appropriate action if it occurs.
The researchers found that the majority of patients with ITP following vaccination had no pre-existing thrombocytopenia. The median time to onset of ITP was 6 days after vaccination, and the median duration of ITP was 4 weeks. Most patients responded to treatment with corticosteroids or intravenous immunoglobulin (IVIG). The researchers concluded that ITP is a rare but serious adverse event following vaccination, and that prompt diagnosis and treatment are essential for successful outcomes.
Researchers also suggested that healthcare providers should be aware of the potential for ITP and other thrombocytopenia-related events post vaccination, and that patients should be monitored closely for any signs or symptoms of thrombocytopenia. They also recommended that further research should be conducted to better understand the potential link between the vaccines and ITP.
- “While the main concern associated with ITP is bleeding, it may come as a surprise that ITP is also associated with a 20% increased risk for blood clots,†Redwood said, pointing to a March 8 article by Dr. Robert Bird, director of haematology at Princess Alexandra Hospital in Brisbane, Australia.
Physicians weigh in on how vaccines might lead to blood clots
The AAPS said this could lead to “unintended consequences” and that the long-term effects of the vaccines are unknown.
When a vaccinated person is exposed to the virus, their body will recognize it as a threat and activate its immune response. The body will produce antibodies that are specific to the virus, which will help fight off the infection. This should reduce the severity of symptoms and may even prevent them from developing altogether.
The spike protein is the main component of the virus that allows it to attach to and enter cells. It is also responsible for triggering an immune response in the body, which can lead to inflammation and other symptoms associated with COVID-19.
The FDA is currently reviewing the data and evidence presented by AAPS physicians and scientists. The agency has not yet issued a response to the potential risks associated with mRNA products. In the meantime, the FDA is continuing to monitor the safety of these products and will take appropriate action if necessary.
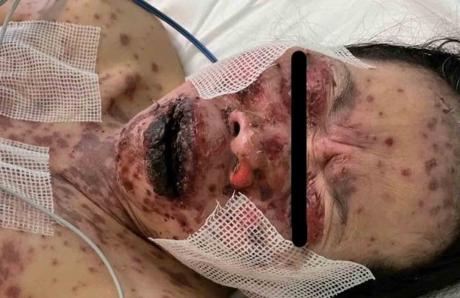
The American Association of Physicians and Surgeons (AAPS) has reported that at least 37 people have developed a rare platelet disorder after receiving the Pfizer or Moderna COVID-19 vaccine. The disorder, known as immune thrombocytopenia (ITP), is characterized by low levels of platelets in the blood, which can lead to excessive bleeding and bruising.
The AAPS also reported that one of those affected was 56-year-old Florida obstetrician Gregory Michael, who developed ITP three days after receiving the Pfizer product and died 15 days later.
The Centers for Disease Control and Prevention (CDC) is currently investigating these cases to determine if there is a causal link between the vaccine and ITP. In the meantime, they are urging healthcare providers to report any cases of ITP following vaccination to their local health department or Vaccine Adverse Event Reporting System (VAERS).
During the interview, Noorchashm discussed his research on blood clots and the potential risks associated with certain vaccines. He also spoke about the need for more transparency in vaccine safety data and urged people to be cautious when considering getting vaccinated. Noorchashm concluded by emphasizing that it is important to weigh the risks and benefits of any medical intervention before making a decision.
vaccines.
It is concerning that the FDA is not taking similar thrombotic complications with Pfizer and Moderna vaccines seriously. It is important for the FDA to investigate all potential side effects of any vaccine, regardless of the manufacturer, in order to ensure the safety of those receiving it.
Scientist warned FDA in December about COVID vaccines and blood clots
- On Dec. 8, 2020, before any COVID vaccines received Emergency Use Authorization in the U.S., J. Patrick Whelan, M.D., Ph.D., wrote the FDA about the potential for vaccines designed to create immunity to the SARS-CoV-2 spike protein “to cause microvascular injury and blood clots throughout the body including the brain, heart, liver and kidneys in ways that were not assessed in the safety trials.â€
However, Whelan argued that the vaccines’ long-term safety and effectiveness remain unknown. He also raised concerns about the potential for adverse reactions to the vaccine, including anaphylaxis, a life-threatening allergic reaction.
- But Whelan cautioned that “it would be vastly worse if hundreds of millions of people were to suffer long-lasting or even permanent damage to their brain or heart microvasculature as a result of failing to appreciate in the short-term an unintended effect of full-length spike protein-based vaccines on other organs.â€
The U.S. Centers for Disease Control and Prevention (CDC) and the Food and Drug Administration (FDA) are investigating these cases to determine if they are related to the vaccine.
Cerebral venous thrombosis (CVT) is a rare but serious condition that occurs when a blood clot forms in the veins that drain blood from the brain. It can be caused by a variety of factors, including infections, medications, and genetic conditions. While CVT has been reported in cases of COVID-19 infection, it is not clear if there is an increased risk for those who receive the vaccine. However, it is important to be aware of the potential risks associated with any medical intervention and to seek medical attention if you experience any symptoms such as headache, confusion, or seizures.
- “The science has progressed rapidly and we are now aware that the spike protein alone, without COVID infection, is capable of traveling through the body and binding to ACE-2 receptors and activating a host of cell-signaling pathways capable of triggering a wide range of adverse events,†Redwood said.
The study found that the spike protein was able to enter the brain and other organs without causing any inflammation or damage. This suggests that S1 could potentially be used as a therapeutic agent for treating neurological diseases, such as Alzheimer’s disease and Parkinson’s disease. The study also showed that S1 could be used to target specific cells in the brain, which could lead to new treatments for neurological disorders.
This could lead to an increased risk of thrombosis and other cardiovascular complications. The authors suggest that this mechanism may explain the high prevalence of cardiovascular complications in COVID-19 patients.
- According to Redwood, it’s “only logical to assume†that when the vaccine creates the identical spike protein that occurs in infection, and which has been identified as the culprit causing a myriad serious and life threatening injuries, “we are going to see these same injuries in individuals who receive the vaccines.â€
He also said that it is important to consider the potential long-term effects of these vaccines, as well as any other potential risks associated with them. Redwood also noted that it is essential to ensure that all vaccine recipients are adequately informed about the risks and benefits of vaccination before they receive the vaccine.
You may also like